As utilities across the country work to address PFAS in drinking water, granular activated carbon (GAC) has become one of the most widely adopted treatment technologies. GAC is effective at adsorbing PFAS, but PFAS are not the only constituents competing for adsorption sites on the carbon.
In many source waters, naturally occurring metals such as iron and manganese, along with organic compounds, can significantly reduce the effectiveness of GAC systems. When these constituents occupy active sites on the carbon, PFAS removal efficiency drops, operational costs rise, and premature media changeouts become more likely.
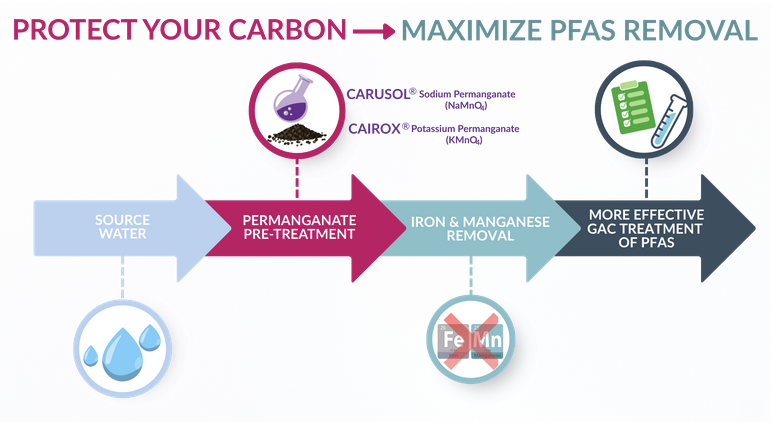
This is where permanganate plays an important, and often overlooked, role.
The Challenge: Protecting GAC Performance in Real-World Water Conditions
GAC does not selectively adsorb PFAS alone. Iron, manganese, and other oxidizable compounds can also bind to the carbon surface, effectively plugging active sites that would otherwise be available for PFAS removal. Over time, this can lead to:
- Reduced PFAS treatment efficiency
- Increased frequency of carbon replacement
- Higher operational and lifecycle costs
- Earlier-than-expected breakthrough
For utilities investing heavily in PFAS treatment infrastructure, protecting GAC performance is critical.
Why Permanganate?
Permanganate has long been recognized as a reliable oxidant for removing iron and manganese. When applied before filtration, permanganate oxidizes these metals into forms that can be removed prior to the GAC system, preventing them from reducing the GAC’s effectiveness.
While other oxidants, such as chlorine, can be used, they often come with limitations:
- Manganese oxidation with chlorine is slow and less effective
- In the presence of ammonia, chlorine demand increases significantly, complicating treatment
- Chlorine performance can be inconsistent under varying water quality conditions
Permanganate offers a more targeted and efficient alternative, particularly in source waters with elevated manganese and/or ammonia.
A Midwest Case Study: Supporting PFAS Treatment with CARUSOL® Sodium Permanganate
A Midwest drinking water facility recently implemented a GAC system to address PFAS concerns. Recognizing the potential impact of iron and manganese on GAC performance, the utility elected to incorporate CARUSOL® sodium permanganate pre-treatment ahead of the carbon system.
The results were clear:
- Iron and manganese were effectively removed by CARUSOL, upstream of GAC
- Carbon active sites were preserved for PFAS adsorption
- GAC life was extended, reducing replacement frequency
- Overall PFAS treatment costs were lowered
By addressing competing constituents before they reached the carbon, the utility was able to maximize the return on its PFAS treatment investment.
A Broader Perspective on PFAS Treatment
PFAS treatment does not exist in isolation. Source water quality, competing contaminants, and upstream treatment decisions all influence system performance and cost. Permanganate is not a PFAS removal technology, but it is a powerful tool that helps PFAS treatment technologies perform as intended.
As utilities continue to evaluate and implement PFAS solutions, considering the supporting role of oxidants like permanganate can make a meaningful difference in both performance and long-term sustainability.

Looking Ahead
This application highlights yet another way permanganate continues to evolve beyond its traditional roles. By protecting downstream treatment assets, permanganate helps utilities address emerging contaminants while managing costs and operational risk.
At Carus, we’re proud to support innovative, practical solutions that help utilities meet today’s challenges and create cleaner, safer environments.

